These three elements also displayed similar chemical reactions, such as vigorously reacting with the members of another triad: chlorine, bromine, and iodine. When the atomic masses of lithium and potassium were averaged together, it was approximately equal to the atomic mass of sodium (22.99). Dobereiner found that the atomic masses of these three elements, as well as other triads, formed a pattern. Triads were based on both physical as well as chemical properties. One such triad was lithium, sodium, and potassium. In 1829, a German chemist, Johann Dobereiner (1780-1849), placed various groups of three elements into groups called triads. In other words, putting elements in separate groups based on how they reacted with other elements. The question was: how?Ī logical way to begin grouping elements together was by their chemical properties. With the ever-increasing number of elements, chemists recognized that there may be some kind of systematic way to organize the elements. As scientific methods improved, the rate of discovery dramatically increased. Several of these, such as copper and lead, had been known since ancient times. Early Attempts to Organize Elementsīy the year 1700, only a handful of elements had been identified and isolated. Chemistry information also needs to be organized so we can see patterns of properties in elements. /periodic-table-of-the-elements-2017--illustration-769723031-5aa02f9b04d1cf00386ccf7c.jpg)
Both approaches organize information so that people can easily find what they are looking for. This method is one of the most widely used ways to organize libraries in the world. This number refers to the Dewey Decimal system, developed by Melvil Dewey in 1876 and used in over 200,000 libraries throughout the world.Īnother system in wide use is the Library of Congress approach, developed in the late 1800s-early 1900s to organize the materials in the federal Library of Congress. The book you are looking for will have a number by the title. If you are looking for a non-fiction publication, you look in a catalog (most likely on a computer these days). If it is a fiction book, you look by author since the fiction materials are filed by the author’s last name. It is important to recognize which numbering system is being used and to be able to find the number of valence electrons in the main block elements regardless of which numbering system is being used.When you go to the library to find a book, how do you locate it? You will come across periodic tables with both numbering systems. In this numbering system, group 1A is group 1 group 2A is group 2 the halogens (7A) are group 17 and the noble gases (8A) are group 18. The elements in this group are also gases at room temperature.Īn alternate numbering system numbers all of the \(s\), \(p\), and \(d\) block elements from 1-18. We will learn the reason for this later, when we discuss how compounds form. These elements also have similar properties to each other, the most significant property being that they are extremely unreactive, rarely forming compounds. This group contains very reactive nonmetal elements. Group 7A (or 17) elements are also called halogens. It is important to recognize a couple of other important groups on the periodic table by their group name. 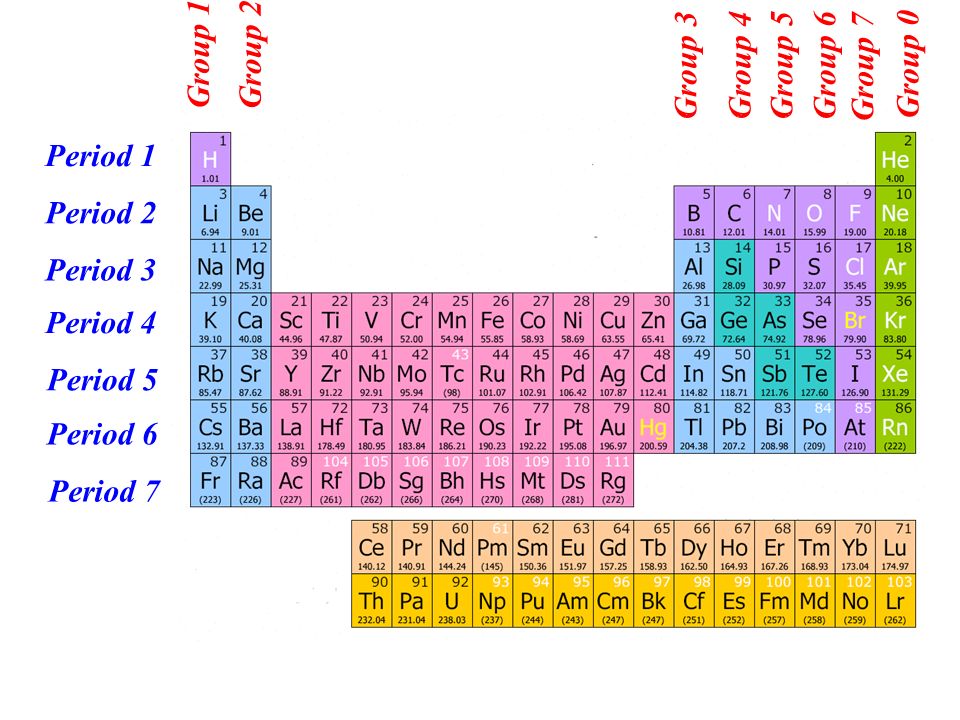
Remember, Mendeleev arranged the table so that elements with the most similar properties were in the same group on the periodic table. The same pattern is true of other groups on the periodic table. Once again, because of their similarities in electron configurations, these elements have similar properties to each other. Group 2A is also called the alkaline earth metals. Although most metals tend to be very hard, these metals are actually soft and can be easily cut. Group 1A is also known as the alkali metals. Because of their similarities in their chemical properties, Mendeleev put these elements into the same group. The elements in 1A are all very reactive and form compounds in the same ratios with similar properties with other elements. This is what causes these elements to react in the same ways as the other members of the family. All of the 1A elements have one valence electron. A group is a vertical column of the periodic table. Remember that Mendeleev arranged the periodic table so that elements with the most similar properties were placed in the same group.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
